OpenText Documentum Content Management for Life Sciences
Ensure GxP-compliant content management in life sciences
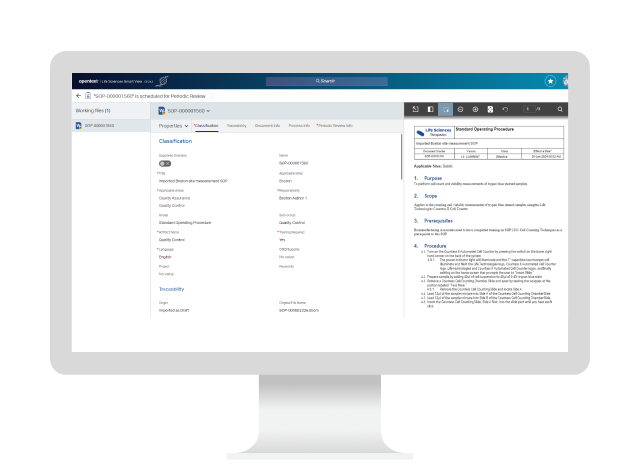
Is your life sciences content ready for the next regulatory inspection?
If you're drowning in documents, siloed systems and processes, and regulatory hurdles, you're not alone. There’s a better way to manage content—faster, smarter, and built for the pace of modern life sciences.

OpenText™ Documentum™ Content Management (CM) for Life Sciences is a GxP-compliant document management system purpose-built for life sciences. It unifies regulated content across clinical, regulatory, quality, and manufacturing domains—keeping critical life sciences content organized and following strict regulations for safety and quality—while enabling secure, cloud-ready collaboration, automated workflows, and seamless system integrations.
Why OpenText Documentum CM for Life Sciences?
Only OpenText combines 25+ years of life sciences expertise with a proven platform trusted by global leaders. Our solution goes beyond compliance, streamlining innovation, enabling speed, and letting you scale in the cloud with confidence.
- 25+ years
industry-proven, GxP-ready
Rely on a solution designed for life sciences with built-in support for 21 CFR Part 11, audit trails, e-signatures, and regulatory standards. - 3.5x
more documents processed monthly
Free up time, cut risk, and make room for real business growth with digital workflows.
Read the customer story - 3
purpose-built life sciences solutions
Let your content move as fast as science by securely handling clinical, regulatory, and quality documents in one system.
Use cases
From clinical trials to commercial launch, teams rely on OpenText Documentum CM for Life Sciences to manage regulated content with speed and precision, simplify compliance, boost productivity, and bring life-saving therapies to market faster.
-
Automate compliance with ICH-compliant filings, using taxonomies, lifecycles, and workflows. Enable editing, merging, and secure archiving with metadata-driven access control through OpenText Documentum CM for Regulatory.
Read the product overview -
-
Support consistent quality, organization, and management of GMP compliant documents—along with secure, traceable printing and tracking—using powerful, controlled templates from OpenText Documentum CM for Quality and Manufacturing.
Read the product overview -
Work smarter by linking to tools like CTMS, LIMS, LMS, QMS, and ERP, so your info stays connected and easy to manage.
-
Share and edit documents securely—whether your team is in the lab or at home—to keep work protected and moving in the right direction.
-
Use pre-built solutions designed for life sciences teams to help you set up quickly and follow important industry rules like IDMP and eTMF.
Key features
Consolidate all life sciences document management in one easy system. From clinical trials to quality and manufacturing or regulatory activities, get the tools you need to stay organized, follow the rules, and work smarter.

GxP compliance framework
Delivers validation support with audit trails, e-signatures, and automated controls to ensure regulatory compliance and reduce inspection risk.

Unified repository
Keeps clinical, regulatory, and manufacturing files together and secure, so you can easily find and manage everything in one spot.

Cloud or on-site deployment
Deploys your way—on-premises or in the cloud—leveraging smart tech like Docker and Kubernetes to make updates and scaling simple.

Ready-made workflows
Helps you get started fast with workflows, templates, and folders built for your industry, and ensures you adhere to global standards.

Automated document checks
Spots missing or wrong documents with helpful charts and reports, and fixes problems quickly before they become issues.

App integration
Links the system to your existing quality, training, and publishing apps, so you don’t have to switch between programs.
Smart tagging and document tracking
Uses built-in rules and metadata tags to easily and consistently manage documents the right way from start to finish.

Scalable content management
Captures, indexes, organizes, stores, tracks, and archives documents securely in a repository built to meet corporate mandates and handle extreme demands, like importing 20 million objects per hour.
How to buy
Explore three scalable, future-ready plans for OpenText Documentum CM for Life Sciences—eTMF, Quality and Manufacturing, and Regulatory —designed to meet the needs of life sciences organizations.
| Swipe to see more | Boost team productivity | Build cross-functional excellence | Bridge across boundaries |
|---|---|---|---|
Simplify long-term data compliance and IT operations and accelerate migration to the cloud.
| Boost team productivity | Build cross-functional excellence | Bridge across boundaries | |
|---|---|---|---|
Add On | |||
Enhance your content services platform with industry and departmental solutions.
| Boost team productivity | Build cross-functional excellence | Bridge across boundaries | |
|---|---|---|---|
Add On | |||
Add On | |||
Add On | |||
Add On | |||
Add On | |||
Understand and action organizational knowledge with AI-powered content management solutions.
| Boost team productivity | Build cross-functional excellence | Bridge across boundaries | |
|---|---|---|---|
Add On | |||
Add On | |||
Use your cloud provider credits to get started
Available directly from OpenText or on your favorite cloud marketplace.

AWS MarketplaceLearn more
Deploy seamlessly on AWS for scalability and global reach

Google Cloud MarketplaceLearn more
Launch on Google Cloud for AI-driven speed and insights

Microsoft MarketplaceLearn more
Run on Azure for secure, enterprise-grade integration
Integrations
Connect your life sciences content with these integrations.
Accelerate the value of OpenText Documentum Content Management for Life Sciences
Add-ons
Maximize your investment in OpenText Documentum CM for Life Sciences with powerful extensions.
-
Find business content faster and eliminate manual tasks with an AI-powered content assistant
Reach new heights in productivity with a secure AI content assistant
-
Simplify cloud migration with efficient application decommissioning
Reduce IT costs and accelerate the move to a modernized, cloud-based architecture with a scalable, economical, and compliant archiving solution
-
Gain insights with knowledge discovery for any repository or format
Easily find and understand your unstructured data using AI and machine learning
-
Transform paper and digital content into actionable insights
Ensure accurate information is available when and where it’s needed, using AI and machine learning to automate content processes and securely route information to the right users and systems
Integrations
Integrate the systems you use every day with your enterprise content management tools.
Deployment
Explore scalable and flexible deployment options for global organizations of any size.
-
Get AI ready with OpenText Thrust APIs
API from OpenText Developer Cloud
Services
Accelerate digital transformation with guidance from certified experts.
-
Modernize your information management with certified experts
Professional Services
-
Turn support into your strategic advantage
Support Services
-
Meet business goals with expert guidance, managed services, and more
Customer Success Services
-
Free up your internal teams with expert IT service management
Managed Services
Partners
OpenText helps customers find the right solution, the right support, and the right outcome.
Global system integrators (GSIs)
These GSIs are trained and certified on OpenText solutions, offering services that enhance the value of stand-alone solutions.
Training
OpenText Learning Services offers comprehensive enablement and learning programs to accelerate knowledge and skills.
-
OpenText Documentum Content Management for Life Sciences Learning Paths
This course provides a comprehensive functional overview of the OpenText Documentum CM for Quality and Manufacturing solution. You will learn about workflows, lifecycles, audits, and search features
Communities
Explore our OpenText communities. Connect with individuals and companies to get insight and support. Get involved in the discussion.
-
A private group designed to give customers, partners, and employees a safe place to learn, share, or submit questions about OpenText Documentum CM
OpenText Documentum CM LinkedIn user group
Premium Support
Optimize the value of your OpenText solution with dedicated experts who provide mission-critical support for your complex IT environment.
Certifications
Ensure your enterprise content management software meets global compliance standards to secure data, manage records, and protect content in regulated environments.
-
Manage patient and clinical records in line with HIPAA regulations using secure access, audit trails, and compliant storage
HIPAA
-
Confirm electronic records and signatures are secure, reliable, and legally equivalent to paper records as determined by the FDA
21 CFR Part 11
-
Benefit from certified security practices that align with ISO 27001 and SOC 2 standards for data confidentiality, integrity, and availability
ISO and SOC 2 compliance
-
Store and manage financial data securely with support for Payment Card Industry Data Security Standard (PCI DSS) requirements
PCI DSS
-
Meet quality standards and ensure products are traceable through their development and manufacturing lifecycle as determined by the FDA and EMA
GxP
-
Guarantee enterprise content adheres to GDPR privacy regulations with strong access controls, auditability, and lifecycle management
GDPR
-
Comply with U.S. Department of Defense standards for managing official records, including classification, retention, and disposition
DoD 5015.2
OpenText Documentum Content Management for Life Sciences resources

Managing quality and manufacturing documentation for life sciences
Watch the video
Get rid of costly dead-weight data with information archiving solutions
Watch the video-
OpenText Documentum CM for Life Sciences helps manage documents by keeping them organized and compliant with rules. It speeds up clinical trials, regulatory submissions, and quality control with secure, easy-to-use tools.
-
OpenText Documentum CM for Life Sciences organizes trial documents, checks for errors like missing approvals, and lets teams share files securely. This cuts delays and keeps everything ready for inspections.
-
OpenText Documentum CM for Life Sciences uses templates and workflows to create submission-ready documents fast, helping organization meet FDA and EMA rules with clear audit trails.
-
It manages GMP documents with templates to reduce errors, simplify reviews, and ensure secure, traceable printing for compliance.
-
Yes, it connects with existing systems like CTMS, QMS, LMS, and LIMS. That means your data flows smoothly between systems, and you don’t have to switch back and forth.
-
It lets you securely print controlled copies with watermarks, expiration dates, and access logs, helping meet GxP and audit requirements.
-
No, you can run it on-premises, in your own private cloud, or with a public cloud provider. It’s flexible, so you can choose what works best for your team.

What’s new in OpenText Documentum for Life Sciences solutions
Learn about the latest new functionality and how it can benefit your organization.
Read the blog
What’s new in OpenText Documentum Content Management
Learn about the latest new functionality and how it can benefit your organization.
Read the blogWhat's new in OpenText Documentum Content Management (CM) for Life Sciences 25.4
Watch the webinarOpenText Content Cloud Virtual Summit 2025
Watch the on-demand webinarWhat’s new in OpenText Documentum CM for Life Sciences CE 24.4
Watch the on-demand webinarOpenText Content Cloud Virtual Summit 2025
Watch the on-demand webinarWhat’s new in OpenText Documentum CM for Life Sciences CE 24.4
Watch the on-demand webinar
Take the next step











